- Lexaria's DehydraTECH-nicotine pouch performance to be compared to existing leading brands ON! and Zyn
KELOWNA, BC / ACCESSWIRE / November 1, 2022 / Lexaria Bioscience Corp. (Nasdaq:LEXX) (Nasdaq:LEXXW) (the "Company" or "Lexaria"), a global innovator in drug delivery platforms is pleased to announce that independent review board ("IRB") approval has now been received for human clinical nicotine study NIC-H22-1, and that human dosing will begin soon.
Study NIC-H22-1 is a 36-person human pharmacokinetic ("pk") randomized, double blinded, cross-over study conducted in current cigarette smokers, wherein each person will visit the laboratory to be dosed three times over a period of weeks. During each visit only one oral nicotine pouch will be administered and evaluated: either DehydraTECH-nicotine; On! brand manufactured by Altria; or Zyn brand manufactured by Swedish Match. Predetermined questionnaires for subjective evaluation will be used for each oral nicotine pouch, and blood samples will be taken a total of 8 times per visit to conduct objective evaluations related to quantity of nicotine in blood at various time points, and more. Vital signs such as temperature, blood pressure, heart rate and respiratory rate will also be collected. Subjective evaluations related to throat burn, user experience, gastrointestinal experience and more will be conducted. Lexaria hopes to evidence that processing purified nicotine with DehydraTECH leads to better oral-tissue absorption and reduced negative experiences compared to currently sold brands.
The study had earlier faced certain time extensions due to manufacturing and logistics, those issues since resolved. The study is fully funded from internal company resources. Lexaria will provide further updates and any relevant material findings in due course from this study as they become available.
As reported on October 5, 2021, Lexaria demonstrated in animal study NIC-A21-1 that nicotine oral pouches using DehydraTECH technology were 10x to 20x faster in reaching peak delivery of nicotine to bloodstream than controls. Findings using a DehydraTECH nicotine benzoate formulation relative to a concentration-matched control from that study are shown in the figure below.
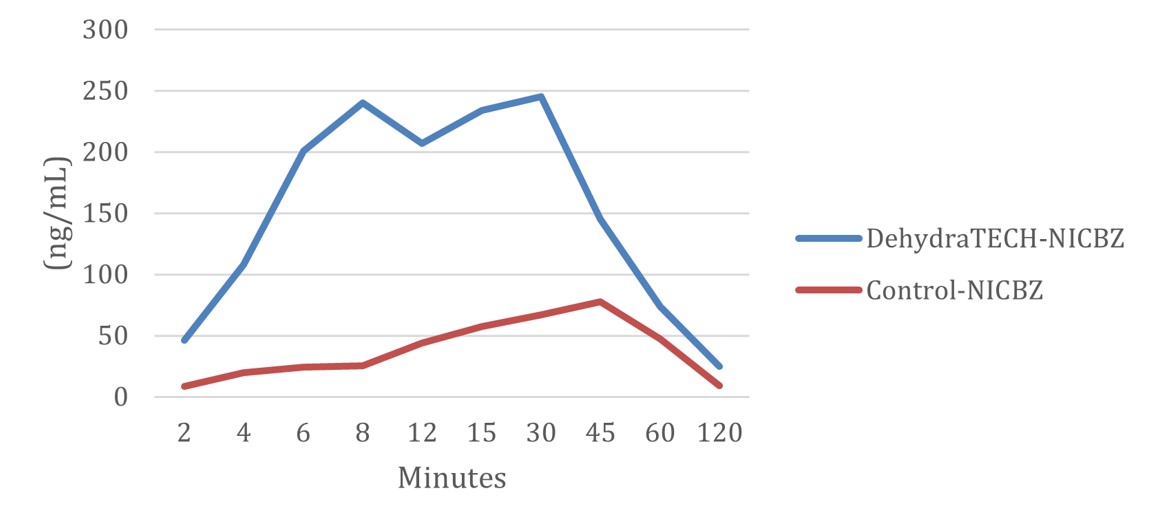
The oral nicotine pouch category is of intense interest to Lexaria and is one of the fastest growing segments of the nicotine industry due in part to its reduced risk health outcomes as noted by the Food and Drug Administration ("FDA"). This delivery method, in the white pouch format specifically, which avoids harmful lung outcomes experienced by smokers or vapers, involves absorption primarily through the buccal tissues of the mouth, of purified nicotine that has been separated from most other harmful compounds in the tobacco leaf. The global market for the oral nicotine pouch category was US$2.33 billion in 2020 and is growing at a rapid CAGR of 30.7% and is expected to reach $21.84 billion in 2027.
As reported on March 8, 2022, Lexaria recently received its first ever patent granted to use DehydraTECH to more efficiently deliver nicotine through buccal tissue absorption. Similar patent filings have been made in the USA and in the EU and Lexaria believes those potential patent awards could support significant competitive advantages in the nicotine white pouch category, as well as other oral nicotine product formats.
ABOUT LEXARIA BIOSCIENCE CORP.
Lexaria Bioscience Corp.'s patented drug delivery technology, DehydraTECH™, improves the way active pharmaceutical ingredients (APIs) enter the bloodstream by promoting more effective oral delivery. Since 2016, DehydraTECH has repeatedly demonstrated the ability to increase bio-absorption with cannabinoids, antiviral drugs, PDE5 inhibitors and more. DehydraTECH has also evidenced an ability to deliver some drugs more effectively across the blood brain barrier. Lexaria operates a licensed in-house research laboratory and holds a robust intellectual property portfolio with 27 patents granted and roughly 50 patents pending worldwide. For more information, please visit www.lexariabioscience.com.
CAUTION REGARDING FORWARD-LOOKING STATEMENTS
This press release includes forward-looking statements. Statements as such term is defined under applicable securities laws. These statements may be identified by words such as "anticipate," "if," "believe," "plan," "estimate," "expect," "intend," "may," "could," "should," "will," and other similar expressions. Such forward-looking statements in this press release include, but are not limited to, statements by the company relating the Company's ability to carry out research initiatives, receive regulatory approvals or grants or experience positive effects or results from any research or study. Such forward-looking statements are estimates reflecting the Company's best judgment based upon current information and involve a number of risks and uncertainties, and there can be no assurance that the Company will actually achieve the plans, intentions, or expectations disclosed in these forward-looking statements. As such, you should not place undue reliance on these forward-looking statements. Factors which could cause actual results to differ materially from those estimated by the Company include, but are not limited to, government regulation and regulatory approvals, managing and maintaining growth, the effect of adverse publicity, litigation, competition, scientific discovery, the patent application and approval process, potential adverse effects arising from the testing or use of products utilizing the DehydraTECH technology, the Company's ability to maintain existing collaborations and realize the benefits thereof, delays or cancellations of planned R&D that could occur related to pandemics or for other reasons, and other factors which may be identified from time to time in the Company's public announcements and periodic filings with the US Securities and Exchange Commission on EDGAR. The Company provides links to third-party websites only as a courtesy to readers and disclaims any responsibility for the thoroughness, accuracy or timeliness of information at third-party websites. There is no assurance that any of Lexaria's postulated uses, benefits, or advantages for the patented and patent-pending technology will in fact be realized in any manner or in any part. No statement herein has been evaluated by the Food and Drug Administration (FDA). Lexaria-associated products are not intended to diagnose, treat, cure or prevent any disease. Any forward-looking statements contained in this release speak only as of the date hereof, and the Company expressly disclaims any obligation to update any forward-looking statements or links to third-party websites contained herein, whether as a result of any new information, future events, changed circumstances or otherwise, except as otherwise required by law.
INVESTOR CONTACT:
George Jurcic - Head of Investor Relations
ir@lexariabioscience.com
Phone: 250-765-6424, ext 202
SOURCE: Lexaria Bioscience Corp.
View source version on accesswire.com:
https://www.accesswire.com/723196/Lexaria-Receives-Independent-Review-Board-Approval-for-its-Upcoming-Human-Oral-Nicotine-Study













